Last Updated : December 9, 2019
Details
Project Status:
Completed
Project Line:
Horizon Scan
Project Sub Line:
Emerging Health Technologies
Project Number:
EH0076-000
Summary
- Atherosclerotic cardiovascular disease (ASCVD) remains one of the leading causes of death in Canada. Cholesterol, specifically low-density lipoprotein cholesterol (LDL-C), is a major risk factor for cardiovascular disease (CVD) and is thereby targeted to reduce the likelihood of a cardiovascular event, such as a myocardial infarction (MI) and stroke.
- Inclisiran, first developed by Alnylam Pharmaceuticals, Inc. (Cambridge, Massachusetts, US) then by The Medicines Company (Parsippany, New Jersey, US), is a small interfering ribonucleic acid (siRNA) molecule being investigated for the treatment of hypercholesterolemia.
- ORION-1 was a phase II, double-blind, placebo-controlled, multi-centre, randomized controlled trial of 501 patients. Patients were included in the trial if they had a history of ASCVD or were at high risk of ASCVD. The treatment arms were administered 200 mg, 300 mg, or 500 mg of inclisiran on day 1, or 100 mg, 200 mg, or 300 mg of inclisiran on days 1 and 90. The comparator was either placebo on day 1 or placebo on days 1 and 90. The primary end point was percentage change in LDL-C at day 180 from baseline.
- The ORION-1 study demonstrated that inclisiran, administered at various doses and intervals, compared with placebo, resulted in a statistically significant reduction in LDL-C levels (P < 0.001 for all comparisons versus placebo). The greatest reduction in LDL-C levels was obtained with the 300 mg dose of inclisiran given at days 1 and 90 with a 52.6% (95% confidence interval [CI]: –57.1 to –48.1) reduction at day 180 compared with baseline, and a mean absolute reduction in LDL-C levels of 1.66 (standard deviation 0.54) mmol/L. Results from the ORION-1 trial provided the necessary data to make a decision regarding the dosing regimen to be used in subsequent phase III trials, in particular the ORION-11 phase III trial.
- The ORION-11 study was a phase III international, multi-centre, and double-blind trial which randomized 1,617 participants (87% with established ASCVD) to inclisiran 300 mg (n = 810) or placebo (n = 807). An initial inclisiran dose of 300 mg given subcutaneously was administered at day 1, day 90, and then every six months for two doses, that is at days 270 and 450. The mean baseline LDL-C level was 2.8 mmol/L (inclisiran) and 2.7 mmol/L (placebo); 96% of participants were on high-dose statin therapy. There was a 50% time-averaged reduction in LDL-C levels from day 90 to day 540 (P < 0.00001). Pre-specified exploratory cardiovascular composite end point (cardiac death, cardiac arrest, MI, or stroke) occurred in 7.8% of inclisiran treated patients versus 10.3% of patients on placebo; this lower rate was mainly driven by a reduction in MI and stroke. With respect to adverse effects, 4.69% of patients on inclisiran reported an injection site reaction, compared with 0.5% of patients on placebo. All reactions were transient. There was no evidence of liver, kidney, muscle, or platelet toxicity.
- Inclisiran may be an option in the future as a cholesterol-lowering medication, where it would likely be used in patients who are unable to achieve their LDL-C targets despite maximally tolerated statin therapy or who are intolerant to statin therapy. However, results from the inclisiran cardiovascular outcome trial (ORION-4), are needed to confirm its efficacy in reducing CVD and its long-term safety.
- Inclisiran is not yet approved by any regulatory authority, but its ORION clinical development program identifies the year 2021 as the goal to reach worldwide markets.
Methods
These bulletins are not systematic reviews and do not involve critical appraisal or include a detailed summary of study findings. Rather, they present an overview of the technology and available evidence. They are not intended to provide recommendations for or against a technology.
Literature Search Strategy
A limited literature search was conducted by an information specialist on key resources including MEDLINE, Embase, PubMed, the Cochrane Library, the University of York Centre for Reviews and Dissemination databases, the websites of Canadian and major international health technology agencies, as well as a focused Internet search. The search strategy was comprised of both controlled vocabulary, such as the National Library of Medicine’s MeSH (Medical Subject Headings) and keywords. The main search concepts were inclisiran and hyperlipidemia. No filters were applied to limit the retrieval by study type. Where possible, retrieval was limited to the human population. The search was also limited to English language documents published between January 1, 2014 and June 7, 2019. Internet links were provided, where available. Alerts were run until September 2019.
Study Selection
One author screened the literature search results and reviewed the full text of all potentially relevant studies. Studies were considered for inclusion if the intervention was inclisiran and hyperlipidemia. Conference abstracts and grey literature were included when they provided additional information to that available in the published studies.
Peer Review
A draft version of this bulletin was reviewed by one clinical expert.
Background
CVD is the second leading cause of death in Canada, accounting for more than 53,000 deaths in 2017.1 It affects approximately 2.4 million, or 1 in 12, Canadians based on 2012-2013 statistics. CVD creates great economic burden on the health care system, costing more than $21 billion annually.2,3
CVD is most commonly characterized by the buildup of plaque on the inner walls of arteries causing a narrowing of the arteries and an obstruction in blood flow. This is referred to as atherosclerosis and it can lead to MI, stroke, and heart failure.3
The plaque that accumulates on the artery walls consists mostly of fat and cholesterol, among other substances. Cholesterol, although crucial in its roles in hormone synthesis and cell membrane integrity, can be deleterious to cardiovascular health if there are abnormal amounts. Dyslipidemia refers to abnormalities in the amount of lipids, where either total cholesterol, low-density lipoprotein cholesterol (LDL-C) or triglycerides are elevated, or high-density lipoprotein cholesterol (HDL-C) is low.4
Hypercholesterolemia, a common type of dyslipidemia, is characterized by elevated levels of LDL-C. It is known to be directly associated with an increased risk of ASCVD. LDL-C can increase atherosclerosis by transporting cholesterol through the blood stream and depositing it in the walls of arteries causing plaque buildup.5 Elevated LDL-C levels can be caused by genetic factors, environmental factors, or secondary to underlying disease.4 Based on 2012-2013 statistics, 19% of the Canadian population between the ages of 18 years and 79 years have elevated LDL-C levels, deemed as ≥ 3.5 mmol/L.5 Familial hypercholesterolemia (FH) is the most prevalent genetic disorder that causes early onset CVD. Approximately 1 in 250 Canadians have FH, which translates to more than 145,000 people in Canada; however, this is likely an underestimate because many cases go undiagnosed. This autosomal dominant genetic disease is caused by a defect in one of the genes related to LDL-C catabolism. An individual with FH is either a homozygote or heterozygote, of which homozygotes are more severely affected with greater LDL-C levels and cardiovascular consequences.6,7
The backbone of therapy for hypercholesterolemia aims at reducing lipid LDL-C levels, thereby reducing plaque formation and consequently, CVD morbidity and mortality. A meta-analysis showed that there is a 22% reduction in CVD events for every 1 mmol/L reduction in LDL-C levels.8 LDL-C lowering therapies include HMG-CoA reductase inhibitors (statins), which are the cornerstone for treating hypercholesterolemia, as well as ezetimibe, proprotein convertase subtilisin-kexin type 9 (PCSK9) inhibitors (evolocumab and alirocumab), bile acid-sequestering resins, and lomitapide9, though the latter drug is limited to use in patients with homozygous FH (HoFH).10 Although there are numerous lipid-lowering drugs available, there is considerable interindividual variability in responses and intolerances to therapy; therefore, some patients remain at suboptimal LDL-C levels. Patients with pre-existing CVD, or with FH are at the highest risk of insufficient reduction in LDL-C levels, of whom it has been shown that less than 50% achieve the target goals with statin therapy.11 These high-risk patients present an unmet need for new cholesterol reducing therapies.
The Technology
The discovery of PCSK9 in 2003 sparked the development of lipid-lowering drugs with a different mechanism of action to the other medications available on the market. PCSK9 is an enzyme predominantly made in the liver; it increases the level of LDL-C in the blood stream. It does so by binding to LDL receptors present on hepatic cells and causing their lysosomal degradation. These LDL receptors normally bind LDL-C and remove it from circulation; therefore, reduced amounts of LDL receptors would cause greater plasma LDL-C levels. This endogenous process was exploited to make PCSK9 inhibitors, which are capable of markedly decreasing LDL-C levels and reducing CVD risk.8,12
Inclisiran (ALN-PCSsc, ALN-60,212) works by targeting the PCSK9 enzyme; however, as opposed to PCSK9 inhibitors, which are antibodies that inhibit circulating PCSK9, inclisiran is a chemically synthesized small interfering RNA (siRNA) molecule that reduces the production of PCSK9 through gene silencing. It consists of two nucleotide strands conjugated to the ligand, triantennary N-acetylgalactosamine (GaINAc). This ligand targets the drug to the liver by binding specifically to the asialoglycoprotein receptors (ASGPR), which are almost exclusively expressed on hepatocytes. Once bound, inclisiran is rapidly taken in by the cell where it then binds to the RNA-induced silencing complex (RISC) as well as the messenger RNA (mRNA) encoding PCSK9. The siRNA RISC complex cleaves the PCSK9 mRNA, preventing the synthesis of the PCSK9 protein. One siRNA RISC complex can do this degradation process with multiple PCSK9 mRNA. Without PCSK9 enzymes, there will be no lysosomal degradation of the LDL receptor, so LDL-C will bind the receptor, be recycled in the hepatocyte, and result in a lower serum LDL-C level. This mechanism of action of inclisiran as well as PCSK9’s role in degrading LDL receptors can be seen in Figure 1.8,12
Figure 1: Inclisiran’s Mechanism of Action on PCSK9 and Its Role in Lysosomal Degradation as well as LDL Receptor’s Cholesterol Recycling Action
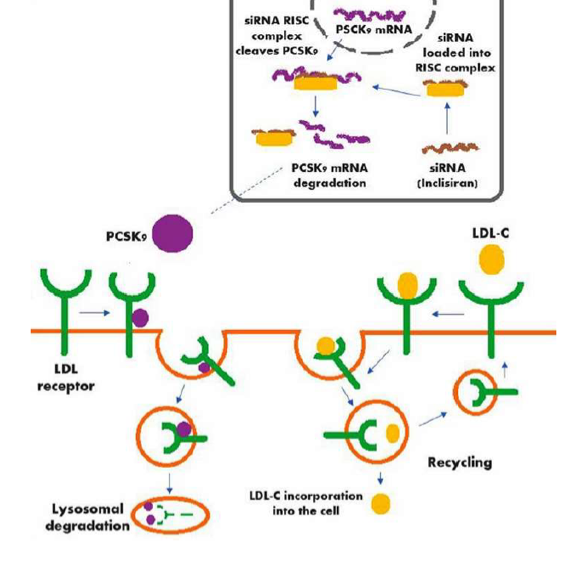
Source : Kosmas CE, Munoz Estrella A, Sourlas A, et al. Inclisiran: a new promising agent in the management of hypercholesterolemia. Diseases. 2018;6(3):13. Copyright 2018 by MDPI AG, Basel, Switzerland. Reproduced according to the Creative Commons License CC BY version 4.0.
Regulatory Status
Inclisiran was first developed by Alnylam Pharmaceuticals, Inc. (Cambridge, Massachusetts, US). Development has now been assumed by The Medicines Company (Parsippany, New Jersey, US).13 One phase I and two phase II trials have been completed. Topline results of two phase III trials were also recently presented while other phase III trials are still ongoing as part of the ORION clinical development program. The anticipated trial completion dates are listed in Table 1.
Inclisiran is not yet approved by any regulatory authority. The anticipated potential date for worldwide market authorization of inclisiran is the year 2021;14 with anticipated regulatory submissions in the US occurring in the fourth quarter of 2019 and, in Europe during the first quarter of 2020.15
Administration and Cost
The dosage and dosing administration pattern established in the phase II ORION-1 trial and being used in ORION phase III trials is one subcutaneous dose of 300 mg inclisiran at days 1 and 90 and then every 180 days.14
The cost of inclisiran is not yet available, given that it is still in clinical development. Hence, neither the Canadian price nor health care system-based economic analyses were identified for inclisiran. Of interest, it has been reported that the manufacturer recently suggested that inclisiran may be priced lower than the currently commercially available PCSK-9 inhibitors.16
Target Population
As previously stated, use of inclisiran is intended for patients with hypercholesterolemia to reduce their LDL-C level. Anticipating the higher price range of this drug, it would most likely be used in high-risk ASCVD patients who either have established intolerance to statin therapy or are unable to reach LDL-C target levels despite use of maximally tolerated statin doses.14,17 Similarly to recently reported data for currently prescribed PCSK9 inhibitors, evolocumab and alirocumab, it may be anticipated that inclisiran could be of greatest benefit to individuals with aggressive coronary artery disease, for example those with recurrent MI, polyvascular, or multivessel disease.18,19
Inclisiran may also be used for the treatment of patients with FH when they are intolerant to or have reached maximum doses of statins. These patients are more at risk for CVD; their hypercholesterolemia is often harder to treat and to get to target levels. Of note, patients with heterozygous FH have been shown to have two-fold higher LDL-C levels compared with unaffected relatives. These patients have a significant 3.6-fold greater risk of CVD14,17; it has been reported that patients with this FH mutation and an LDL-C > 5.0 mmol/L have a 17-fold higher risk of CVD, compared with a five-fold increase in patients without the mutation.20 Statins reduce their CVD morbidity and mortality; however, as mentioned previously less than 50% of patients achieve the lipid target goals with maximally tolerated statin doses. Therefore, this high-risk population might benefit from the release of a new lipid-lowering drug.14,17
Current Practice
Based on the 2016 Canadian Cardiovascular Society and the 2018 American College of Cardiology guidelines, individuals 40 years of age and older or with a risk factor should be screened for their CVD risk. These risk factors include diabetes, atherosclerosis, abdominal aortic aneurysm, active smoking, hypertension, signs of dyslipidemia (arcus cornea, xanthoma, xanthelasma), a family history of premature CVD or dyslipidemia, chronic kidney disease, obesity, inflammatory bowel disease, HIV infection, erectile dysfunction, hypertensive disease during pregnancy, and chronic obstructive pulmonary disease. Screening largely entails performing a non-fasting lipid test, unless the individual has a history of triglyceride levels > 4.5 mmol/L, in which case a fasting lipid level is measured. The levels of total cholesterol, LDL-C, apolipoprotein B (apoB) and non-HDL-C (total cholesterol minus HDL-C) alongside a modified Framingham Risk Score (FRS) or Cardiovascular Life Expectancy Model are assessed to estimate a patient’s cardiovascular risk. An FRS estimates an individual’s 10-year CVD risk and categorizes patients into low, intermediate, and high-risk of CVD. Depending on the level of risk, pharmacological therapy may be recommended; however, this is always in conjunction with lifestyle modifications, which remain the cornerstone for primary and secondary CVD prevention. These health behaviour modifications include maintaining a healthy diet, regular physical activity, smoking cessation, and limiting alcohol consumption.9,21
Most individuals with a modified FRS of < 10%, deemed as low-risk for CVD, do not require pharmacological therapy. Whereas, patients with a high risk of CVD, deemed as having “statin-indicated conditions,” and those with a modified high FRS of > 20% should be prescribed a statin to reduce their CVD risk. Patients with an intermediate CVD risk, which is a modified FRS between 10% and 20%, can be considered for statin therapy depending on their risk factors, or LDL-C, apoB, or non-HDL-C levels. Statin-indicated conditions include patients with clinical atherosclerosis, abdominal aortic aneurysm, most chronic kidney disease, diabetes mellitus, or an elevated LDL-C level. Statins are the mainstay of treating high cholesterol and reducing CVD risk because they can greatly lower LDL-C levels, with reductions ranging from 30% to more than 50%. The optimal lipid levels that are targeted with therapy is an LDL-C of < 2.0 mmol/L, a greater than 50% reduction in LDL-C, an apoB of < 0.8 g/L, or a non-HDL-C of < 2.6 mmol/L. If these targets are not achieved on maximally tolerated doses of a statin then ezetimibe, bile acid-sequestering resins or PCSK9 inhibitors can be added-on to statin therapy. These drugs can also be used if patients are intolerant to statins.9,21 Statin intolerance is ill-defined; however, most experts consider this to be when a patient has tried at least two statins and both have caused intolerable symptoms, most commonly being myopathy, upon rechallenge.22
The management of FH follows the same treatment flow. For this condition, there is a new drug, lomitapide, that is commercially available as adjunctive therapy.6
Summary of the Evidence
The ORION clinical development program for inclisiran includes phase I trials, such as ORION-6 and ORION-7, which examine the pharmacokinetics and safety of inclisiran in hepatic and renal impairment, respectively.14,23 Phase II and phase III trials, which examine the efficacy and safety of inclisiran in larger populations, are the focus of this report (Table 1).
Table 1: Summary of Phase II and III Inclisiran Clinical Trials14,23-32
| Trial | NCT | Clinical Phase and Primary End Point | Population Selected and Sample Size | Actual or Estimated Date of Trial Completion |
|---|---|---|---|---|
| ORION-1 | NCT02597127 | Phase II; LDL-C lowering | ASCVD, ASCVD RE or HeFH N = 501 |
June 2017 |
| ORION-2 | NCT02963311 | Phase II; LDL-C lowering | HoFH N= 4 |
October 2018 |
| ORION-3 | NCT03060577 | Phase II; LDL-C lowering (extension of ORION-1) | ASCVD, ASCVD RE or HeFH N = 490 |
August 2018a January 2022b |
| ORION-4 | NCT03705234 | Phase III; CVOT | ASCVD or ASCVD RE N = 15,000 |
December 2024a December 2049b |
| ORION-5 | NCT03851705 | Phase III; LDL-C lowering | HoFH N = 45 |
June 2021 |
| ORION-8 | NCT03814187 | Phase III; LDL-C lowering (extension of ORION -9, ORION-10, ORION-11) | ASCVD, ASCVD RE or HeFH N = 3,700 |
August 2023a December 2023b |
| ORION-9 | NCT03397121 | Phase III; LDL-C lowering | HeFH N = 482 |
September 2019 |
| ORION-10 | NCT03399370 | Phase III; LDL-C lowering | ASCVD N = 1,561 |
October 2019a December 2019b |
| ORION-11 | NCT03400800 | Phase III; LDL-C lowering | ASCVD or ASCVD RE N = 1,617 |
July 2019a August 2019b |
a Actual or estimated primary outcome measure completion date.
b Estimated study completion date of all outcomes and safety data.
ASCVD = atherosclerotic cardiovascular disease; ASCVD RE = atherosclerotic cardiovascular disease risk equivalents (such as type 2 diabetes, FH, or FRS > 20%); CVOT = cardiovascular outcomes trial; HeFH = heterozygous familial hypercholesterolemia; HoFH = homozygous familial hypocholesterolemia; FH = familial hypocholesterolemia; FRS = Framingham Risk Score; LDL-C = low-density lipoprotein cholesterol; NCT = National Clinical Trial identifier.
In addition to the clinical trials in Table 1, the ORION clinical development program has four additional phase II or phase III trials in the planning stages, including ORION-13, ORION-16, ORION-19, and ORION-20.23 However, due to the lack of information on these future studies, they are not included in Table 1.
ORION-1 Trial
The ORION-1 study is the only trial of the ORION clinical development program for which full results have been published to date. This trial examined the effects of different doses and dosing intervals of inclisiran, as well as its safety and efficacy in lowering LDL-C levels.33 This randomized, double-blind, placebo-controlled, phase II multicenter dose-finding trial included 501 patients who were 18 years of age or older. The study centers were in North America and Europe, so most patients were of European descent. The patients were required to have either an LDL-C level of ≥ 1.8 mmol/L and a history of ASCVD or an LDL-C level of ≥ 2.6 mmol/L with an ASCVD risk equivalent (ASCVD RE) (such as type 2 diabetes or FH). Patients on statins were required to be receiving maximum tolerated doses and be taking a stable dose for at least 30 days. The use of a PCSK9 inhibitor was an exclusion criterion from the study. All patients continued taking their usual lipid-lowering therapies, such as statin with or without ezetimibe, with no changes in medication or dose for the duration of the trial. A total of 73% of patients were taking statins, while 31% were taking ezetimibe, with or without a statin. Patients were randomized to one of eight study groups, these being either placebo or 200 mg, 300 mg, or 500 mg of inclisiran administered on day 1, or placebo or 100 mg, 200 mg, or 300 mg of inclisiran administered on days 1 and 90.24,33
The primary efficacy end point, measured as the least squares mean, was the percentage change from baseline in LDL-C levels at day 180. The secondary efficacy end points included percentage change in serum PCSK9 levels, lipid measures and high-sensitivity C-reactive protein levels at day 180, as well as these measures and LDL-C levels at other time points up to 210 days, or up to 360 days if extended follow-up was necessary. Extended follow-up was done if a patient’s LDL-C level did not return to within 20% of the starting level. A total of 483 patients completed the study and were included in the modified intention-to-treat analysis.33
Reduced LDL-C levels were observed as early as 14 days after the first injection of inclisiran, and by day 180 the percentage of reductions were statistically significantly better compared with the placebo group.33 The study groups with the single dose regimen of inclisiran had reductions in LDL-C levels of 27.9% to 41.9% at day 180, compared with a 2.1% increase in the placebo group (P < 0.001 for all inclisiran doses for the primary efficacy end point, that is the percentage reduction from baseline in LDL-C levels at day 180). In these treatment groups, LDL-C levels were still reduced at day 240 compared with baseline, with reductions ranging between 28.2% and 36.6%.33
The inclisiran groups using two-doses regimens showed reductions in LDL-C levels ranging from 35.5% to 52.6% at day 180. The greatest reduction in LDL-C levels was obtained in the study arm that received two doses of 300 mg of inclisiran, with a mean reduction of 52.6% from baseline and a mean absolute reduction in LDL-C levels of 1.66 (standard deviation 0.54) mmol/L. Among these participants, 54% showed reductions in LDL-C levels of 50% or more. The placebo group part of the two-dose regimen showed a mean increase of 1.8% in LDL-C levels from baseline (P < 0.001 for all comparisons versus placebo); there was also considerable interpatient variability in response.33
Overall, results from the ORION-1 trial provided the necessary data to make a decision regarding the dosing regimen to be used in subsequent phase III trials, in particular the ORION-11 phase III trial.
Preliminary results of two other trials of the ORION program were recently made public, that is the ORION-2 pilot study and the ORION-10 trial. A short description is provided below.
ORION-2 Pilot Study
The ORION-2 study was a phase II trial that served as pilot for a larger phase III trial of inclisiran in patients with HoFH, that is the ORION-5. It was an open-label, single-arm, multi-centre pilot study in HoFH patients receiving high-intensity statin therapy plus ezetimibe. Inclisiran was administered at a dose of 300 mg subcutaneously on days 1 and 90 (if serum PCSK9 level reduction at days 60 or 90 was < 70% compared with baseline). The primary efficacy end point was per cent change from baseline in LDL-C levels on days 90 and 180; secondary efficacy end points included changes in other lipid and lipoprotein parameters and PCSK9 levels. Four HoFH patients with genetically confirmed mutations in both LDL receptor alleles were enrolled. Three patients achieved durable LDL-C level lowering; these reductions ranged between 11.7% and 33.1% on day 90 and between 17.5% and 37% on day 180. All patients also achieved reductions in PCSK9 and apoB levels on days 90 and 180. The patient who did not have a reduction in LDL-C levels had a history of minimal response to both evolocumab and alirocumab. No drug-related adverse events or injection site reactions were reported. Authors concluded that the addition of inclisiran to high-intensity statin therapy plus ezetimibe produces durable LDL-C level lowering in HoFH patients.34
ORION-10 Trial
In a recent press release, The Medicines Company reported that inclisiran met all primary and secondary end points in patients enrolled in the phase III ORION-10 study. In this trial, 1,561 patients with ASCVD and increased LDL-C levels, despite maximum tolerated dose of LDL-C-lowering therapy, were enrolled from 145 US sites. A dose of 300 mg of inclisiran given subcutaneously was evaluated. The primary end points were percentage change in LDL-C levels to day 510 and time-adjusted percentage change in LDL-C levels after day 90 and up to day 540. Secondary end points included the mean absolute change in LDL-C levels at day 510 and the time-adjusted absolute change in LDL-C levels from day 90 and up to day 540, as well as percentage change from baseline to day 510 in PCSK9, total cholesterol, apoB, and non-HDL-C levels. The press release states that inclisiran showed a safety, efficacy and tolerability profile that was at least as favourable as that found in the ORION-11 trial; no treatment-related liver or renal laboratory abnormalities were reported.31,35
ORION-11 Trial15,16,32,36
Full topline results of phase III trial ORION-11 were presented in September 2019 at the European Society of Cardiology’s Congress 2019. This study was an international, placebo-controlled, double-blind, and randomized clinical trial. It evaluated the efficacy, safety, and tolerability of inclisiran in 1,617 patients with ASCVD or ASCVD RE and elevated LDL-C levels despite maximum tolerated dose of statin therapy (with or without ezetimibe). Patients were recruited from 70 sites located in seven countries; there were no US sites. In this study, participants received an initial dose of inclisiran 300 mg subcutaneously, followed by another similar dose at three months and then every six months, that is at days 1, 90, 270, and 450.
The primary end points were percentage change in LDL-C levels from baseline to day 510 and time-adjusted percentage change in LDL-C levels from baseline after day 90 and up to day 540. As for secondary end points, these included the mean absolute change in LDL-C levels at day 510 and the time-adjusted absolute change in LDL-C levels from day 90 and up to day 540, as well as percentage change from baseline to day 510 in PCSK9, total cholesterol, apoB, and non-HDL-C levels.
Patients enrolled in the ORION-11 trial were at high-risk of ASCVD; 87% of patients had been diagnosed with ASCVD while 13% had been diagnosed with ASCVD RE. A balanced randomization between the inclisiran (n = 810) and placebo (n = 807) groups was reported. The mean age of study patients was 65 years; 28% were women. Of the patients with ASCVD RE, 7% had a 10-year ASCVD risk > 20% and 1.7% had heterozygous FH. The mean baseline LDL-C level was 2.8 mmol/L in the inclisiran arm and 2.7 mmol/L in the placebo arm. A high proportion of patients (96%) were taking statins at study entry; 90% of these patients were on high-intensity regimens. Approximately 6% and 8 % of patients in the intervention and placebo arms were on ezetimibe, respectively.
Results for the primary end point of per cent change in LDL-C level from baseline to day 510 included a 49% reduction with inclisiran versus a 4% increase with placebo, leading to a between-group difference of 54% (P < 0.00001). For the second primary end point; that is time-averaged per cent change in LDL-C level from day 90 to day 540, a between-group difference of 50% (P < 0.00001) was reported. It has been commented that the latter measure, which looks at the average LDL-C level reduction over the whole treatment period, may be the more important end point given inclisiran is a long-acting medication with extended dosing intervals.
Results for prespecified exploratory cardiovascular end points were also reported. The rate of the composite end point (cardiac death, cardiac arrest, MI, or stroke) was numerically lower in the inclisiran group (7.8%, n = 63) than in the placebo group (10.3%, n = 83). A similar pattern was reported for individual cardiovascular end points, that is the MI (fatal or nonfatal): inclisiran = 1.2% and placebo = 2.7%; and stroke (fatal or nonfatal) inclisiran = 0.2% and placebo = 1.0%. The rate of cardiovascular death was similar between treatment arms (inclisiran = 1.1% and placebo = 1.2%).
With respect to adverse effects, a higher rate of injection site reactions was observed in the inclisiran group (4.69%, n = 38) than in the placebo group (0.5%, n = 4). Among the 38 patients who had an injection site reaction while taking inclisiran, the reaction was classified as mild in two patients and as moderate in 15 patients. None of these reactions were persistent.
A similar rate of serious adverse effects was reported in both groups with 22.3% of patients on inclisiran and 22.5% of patients on placebo experiencing at least one serious treatment-emergent adverse event. The incidence of deaths (1.7% versus 1.9%) and malignancies (2.0% versus 2.5%) was also similar between the inclisiran and placebo groups. No differences were reported between the inclisiran and placebo groups for laboratory test (liver function tests, renal function tests, creatine kinase measurements, and platelet counts).
Concurrent Developments
The following drugs are being developed for the treatment of dyslipidemia:
- Mipomersen, developed by IONIS and Genzyme, is an LDL-C lowering treatment that was approved in the US in 2013 for patients with HoFH. It is an antisense oligonucleotide that inhibits apoprotein B100 synthesis, which is an essential component of LDL. It was shown to reduce LDL-C levels by 28% in patients with FH. Tolerability of this drug is however limited with high rates of injection site reactions, transaminase elevations, hepatic steatosis, and flu-like symptoms.37,38
- Bempedoic acid, currently in phase III trials, is an oral, once daily, medication that reduces cholesterol synthesis in the liver through the inhibition of adenosine triphosphate citrate lyase. It has shown reductions in LDL-C levels ranging from 13% to 43% in phase I and phase II trials. It is manufactured by Esperion Therapeutics and submissions were filed for FDA and European Medicines Agency approval in February 2019.37-39
- Gemcabene, manufactured by Gemphire Therapeutics, is undergoing phase II and phase III trials to assess its lipid-lowering potential. A study showed that it reduces LDL-C levels by 23% to 28% at varying doses and with the background use of statins.38
- Evinacumab, developed by Regeneron, is an angiopoietin-like protein 3 monoclonal antibody that was shown to reduce LDL-C and triglycerides levels in phase II and phase III trials.38
- RGX-501(AAV8.TBG.hLDLR) is an investigational gene therapy developed by REGENXBIO for the treatment of HoFH. It is currently in phase I and phase II trials.38
Another advanced approach to treating dyslipidemia are vaccines (AFFITOPE AT04A and AT06A) that eliminate circulating PCSK9 through immune activating pathways. There are preliminary results demonstrating the potential lipid-lowering effects of such vaccines. However, it is early in their clinical development as phase I trials are currently ongoing.40
In comparison with these emerging treatments, inclisiran is the only LDL-C lowering drug that is a siRNA molecule. This technology is being explored to treat disorders other than hypercholesterolemia, such as acute hepatic porphyrias and hemophilia with the siRNAs, givosiran, and fitusiran respectively.41,42 Patisiran has recently been approved in Canada for the treatment of familial amyloidotic polyneuropathy as the first RNA interference-based drug.43
Implementation Issues
A potential barrier to the wide adoption of inclisiran could be the cost of the medication. Although its cost is not yet available, it can be anticipated to be relatively high given it provides an alternative approach to the treatment of hypercholesterolemia, that is advanced gene silencing siRNA technology, and may also require a low number of annual injections. That being noted, the manufacturer recently alluded to the possibility that inclisiran may be priced lower than the PCSK-9 inhibitors.16
It remains uncertain if the proposed twice-yearly dosing regimen of inclisiran will provide an advantage in terms of impact on patient’s health outcomes compared with current PCSK9 inhibitors, which are administered subcutaneously once or twice a month. There is nonetheless an unmet need for effective lipid-lowering drugs for high-risk ASCVD, heterozygous FH, and HoFH populations that require further LDL-C levels reduction to reduce their CVD risk.14 It would appear that this new drug product may also be positioned as having an advantage for patients who are not adherent to their medication. This commercial opportunity would be based on the observation that more than 70% of patients with ASCVD or FH currently treated with lipid-lowering drugs are not achieving LDL-C treatment goals, and approximately two-thirds of patients do not adhere to available first-line cholesterol-lowering treatments after one year.15
Additional results from ongoing ORION trials are necessary to further assess the efficacy and safety of inclisiran. The goal for market authorization is the year 2021, which is before the primary completion date of the cardiovascular outcome trial, ORION-4.14 This is an important potential implementation issue for inclisiran since cardiovascular outcomes are preferred to the LDL-C surrogate outcomes for assessing the value of this type of medication.
References
- Leading causes of death, total population, by age group. Table 13-10-0394-01 (formerly CANSIM 102-0561) Ottawa (ON): Statistics Canada: https://www150.statcan.gc.ca/t1/tbl1/en/tv.action?pid=1310039401. Accessed 2019 Jun 20.
- Cardiovascular disease - economic burden of disease Ottawa (ON): Public Health Agency of Canada; 2012: https://www.canada.ca/en/public-health/services/chronic-diseases/cardiovascular-disease/cardiovascular-disease-economic-burden-illness.html. Accessed 2019 Jun 20.
- Report from the Canadian Chronic Disease Surveillance System: heart disease in Canada 2018. Ottawa (ON): Public Health Agency of Canada; 2018: https://www.canada.ca/content/dam/phac-aspc/documents/services/publications/diseases-conditions/report-heart-disease-canada-2018/pub1-eng.pdf. Accessed 2019 Jun 20.
- Talbert RL. Hyperlipidemia. In: DiPiro J, Talbert R, Yee G, Matzke G, Wells B, Posey L, eds. Pharmacotherapy: a pathophysiologic approach. 9th ed. New York (NY): McGraw-Hill Education; 2014.
- Cholesterol levels of adults 2012 to 2013. Ottawa (ON): Statistics Canada: https://www150.statcan.gc.ca/n1/pub/82-625-x/2014001/article/14122-eng.htm. Accessed 2019 Jun 18.
- Brunham LR, Ruel I, Aljenedil S, et al. Canadian Cardiovascular Society position statement on familial hypercholesterolemia: update 2018. Can J Cardiol. 2018;34(12):1553-1563.
- High prevalence of familial hypercholesterolemia (FH) in Canadian population must be addressed: awareness, early screening top priorities to prevent genetic heart disease and death. 2017; https://www.newswire.ca/news-releases/high-prevalence-of-familial-hypercholesterolemia-fh-in-canadian-population-must-be-addressed-awareness-early-screening-top-priorities-to-prevent-genetic-heart-disease-and-death-651821893.html. Accessed 2019 Jun 21.
- Nishikido T, Ray KK. Inclisiran for the treatment of dyslipidemia. Expert Opin Investig Drugs. 2018;27(3):287-294.
- Anderson TJ. 2016 Canadian Cardiovascular Society guidelines for the management of dyslipidemia for the prevention of cardiovascular disease in the adult. Can J Cardiol. 2016;32(11):1263-1282. http://www.ccs.ca/eguidelines/Content/Topics/Dyslipidemia/4.2%20Non-statin%20Therapy.htm?Highlight=hypercholesterolemia. Accessed 2019 Jun 14.
- PrJUXTAPID™ (lomitapide): capsules 5 mg, 10 mg and 20 mg lomitapide (as lomitapide mesylate) microsomal triglyceride transfer protein inhibitor [product monograph]. Toronto (ON): Aegerion Pharmaceuticals (Canada) Ltd.; 2017 Jun 29.
- The Medicines Company. ALN-PCSSC - A placebo-controlled, double-blind, randomized trial to compare the effect of different doses of ALN-PCSSC given as single or multiple subcutaneous injections in subjects with high cardiovascular risk and elevated LDL-C. Protocol no: MDCO-PCS-15-01. Clinicaltrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2016: https://clinicaltrials.gov/ProvidedDocs/27/NCT02597127/Prot_000.pdf. Accessed 2019 Jun 21.
- Kosmas CE, Munoz Estrella A, Sourlas A, et al. Inclisiran: a new promising agent in the management of hypercholesterolemia. Diseases. 2018;6(3):13.
- The Medicines Company and Alnylam Pharmaceuticals announce initiation of phase III clinical trials of inclisiran. 2017; http://investors.alnylam.com/news-releases/news-release-details/medicines-company-and-alnylam-pharmaceuticals-announce. Accessed 2019 Jun 26.
- Stoekenbroek RM, Kallend D, Wijngaard PL, Kastelein JJ. Inclisiran for the treatment of cardiovascular disease: the ORION clinical development program. Future Cardiol. 2018;14(6):433-442.
- The Medicines Company presents results from ORION-11, first phase 3 trial of inclisiran, showing durable and potent lowering of LDL-C with twice-yearly dosing. https://www.biospace.com/article/releases/the-medicines-company-presents-results-from-orion-11-first-phase-3-trial-of-inclisiran-showing-durable-and-potent-lowering-of-ldl-c-with-twice-yearly-dosing/. Accessed 2019 Oct 29.
- Hughes S. ORION-11: 50% reduction in LDL with twice-yearly injection. Medscape 2019; https://www.medscape.com/viewarticle/918130#vp_3. Accessed 2019 Oct 29.
- Ontario Drug Benefit Formulary. Comparative drug index. 2018; https://www.formulary.health.gov.on.ca/formulary/. Accessed 2019 Jun 21.
- Jukema JW, Szarek M, Zijlstra LE, et al. Alirocumab in patients with polyvascular disease and recent acute coronary syndrome: ODYSSEY OUTCOMES trial. J Am Coll Cardiol. 2019;74(9):1167-1176.
- Sabatine M, De Ferrari G, Giugliano R, et al. Clinical benefit of evolocumab by severity and extent of coronary artery disease: analysis from FOURIER. Circulation. 2018;138:756-766.
- Khera AV, Won HH, Peloso GM, et al. Diagnostic yield and clinical utility of sequencing familial hypercholesterolemia genes in patients with severe hypercholesterolemia. J Am Coll Cardiol. 2016;67(22):2578-2589.
- Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol. Circulation. 2018(139):e1082-e1143.
- Baum SJ, Toth PP, Underberg JA, Jellinger P, Ross J, Wilemon K. PCSK9 inhibitor access barriers-issues and recommendations: Improving the access process for patients, clinicians and payers. Clin Cardiol. 2017;40(4):243-254.
- Raal FJ. Inclisiran and the ORION clinical development programme. PCSK9 Forum. https://www.pcsk9forum.org/wp-content/uploads/Inclisiran-and-the-ORION-programme.pdf. Accessed 2019 Jun 18.
- The Medicines Company. NCT02597127: Trial to evaluate the effect of ALN-PCSSC treatment on low density lipoprotein cholesterol (LDL-C) (ORION-1). ClincialTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2015: https://clinicaltrials.gov/ct2/show/NCT02597127 Accessed 2019 Jun 24.
- The Medicines Company. NCT02963311: A study of ALN-PCSSC in participants with homozygous familial hypercholesterolemia (HoFH) (ORION-2). ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2016: https://clinicaltrials.gov/ct2/show/NCT02963311?term=NCT02963311&rank=1. Accessed 2019 Jul 9.
- The Medicines Company. NCT03060577: An extension trial of inclisiran compared to evolocumab in participants with cardiovascular disease and high cholesterol. ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine: https://clinicaltrials.gov/ct2/show/NCT03060577?term=NCT03060577&rank=1. Accessed 2019 Jul 9.
- University of Oxford. NCT03705234: A randomized trial assessing the effects of inclisiran on clinical outcomes among people with cardiovascular disease (ORION-4). ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine: https://clinicaltrials.gov/ct2/show/NCT03705234?term=NCT03705234&rank=1. Accessed 2019 Jul 9.
- The Medicines Company. NCT03851705: A study of inclisiran in participants with homozygous familial hypercholesterolemia (HoFH) (ORION-5). ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2019: https://clinicaltrials.gov/ct2/show/NCT03851705?term=NCT03851705&rank=1. Accessed 2019 Jul 9.
- The Medicines Company. NCT03814187: Trial to assess the effect of long term dosing of inclisiran in subjects with high CV risk and elevated LDL-C (ORION-8). ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2019: https://clinicaltrials.gov/ct2/show/NCT03814187?term=NCT03814187&rank=1. Accessed 2019 Jul 9.
- The Medicines Company. NCT03397121: Trial to evaluate the effect of inclisiran treatment on low density lipoprotein cholesterol (LDL-C) in subjects with heterozygous familial hypercholesterolemia (HeFH) (ORION-9). ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2018: https://clinicaltrials.gov/ct2/show/NCT03397121?term=NCT03397121&rank=1. Accessed 2019 Jul 9.
- The Medicines Company. NCT03399370: Inclisiran for participants with atherosclerotic cardiovascular disease and elevated low-density lipoprotein cholesterol (ORION-10). ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2018: https://clinicaltrials.gov/ct2/show/NCT03399370?term=NCT03399370&rank=1. Accessed 2019 Jul 9.
- The Medicines Company. NCT03400800: Inclisiran for subjects with ACSVD or ACSVD-risk equivalents and elevated low-density lipoprotein cholesterol (ORION-11). ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2018: https://clinicaltrials.gov/ct2/show/NCT03400800?term=NCT03400800&rank=1. Accessed 2019 Jul 9.
- Ray KK, Landmesser U, Leiter LA, et al. Inclisiran in patients at high cardiovascular risk with elevated LDL cholesterol. N Engl J Med. 2017;376(15):1430-1440.
- Raal F, Lepor N, Kallend D, Stoekenbroek R, Wijngaard P, Hovingh GK. Inclisiran durably lowers Ldl-C And Pcsk9 expression in subjects with homozygous familial hypercholesterolaemia: The Orion-2 pilot study. Atherosclerosis. 2019;287:e7.
- Medicines Company’s inclisiran meets all endpoints in ORION-10 trial. Clinical Trials Arena 2019; https://www.clinicaltrialsarena.com/news/inclisiran-orion-10-trial-data/. Accessed 2019 Oct 29.
- American College of Cardiology. ORION-11: substantial LDL-C reduction with twice yearly dosing of novel inclisiran. 2019: https://www.acc.org/latest-in-cardiology/articles/2019/08/28/13/42/mon-230am-orion-11-esc-2019. Accessed 2019 Oct 29.
- Bove M, Cicero AFG, Borghi C. Emerging drugs for the treatment of hypercholesterolemia. Expert Opin Emerg Drugs. 2019;24(1):63-69.
- Hegele RA, Tsimikas S. Lipid-lowering agents. Circ Res. 2019;124(3):386-404.
- Bempedoic acid: a novel agent. Ann Arbor (MI): ESPERION:https://www.esperion.com/pipeline/bempedoic-acid. Accessed 2019 Jul 3.
- Seidah NG, Prat A, Pirillo A, Catapano AL, Norata GD. Novel strategies to target proprotein convertase subtilisin kexin 9: beyond monoclonal antibodies. Cardiovasc Res. 2019;115(3):510-518.
- Lee K, Jang B, Lee YR, et al. The cutting-edge technologies of siRNA delivery and their application in clinical trials. Arch Pharm Res. 2018;41(9):867-874.
- Alnylam presents positive complete results from ENVISION phase 3 study of givosiran, an investigational RNAi therapeutic for the treatment of acute hepatic porphyria. 2019: http://investors.alnylam.com/news-releases/news-release-details/alnylam-presents-positive-complete-results-envision-phase-3. Accessed 2019 Jul 3.
- Health Canada drug product database online query. 2019; https://health-products.canada.ca/dpd-bdpp/index-eng.jsp. Accessed 2019 Jul 3.
About this Document
Authors: Mariana Klinovski, Michel Boucher, Christine Perras, Aleksandra Grobelna
Acknowledgments: CADTH would like to acknowledge the contribution of Ruth McPherson, MD, PhD, FRCPC, FACP, FRSC from the Ottawa Heart Institute for her review of the draft version of this bulletin.
CADTH thanks the external reviewers who kindly provided comments on an earlier draft of this bulletin.
Cite as: Inclisiran: A Small Interfering RNA Molecule for Treating Hypercholesterolemia. Ottawa: CADTH; 2019 Dec. (CADTH Issues in Emerging Health Technologies; Issue 180).
ISSN: 1488-6324
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policymakers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein do not necessarily reflect the views of Health Canada, Canada’s provincial or territorial governments, other CADTH funders, or any third-party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.
Last Updated : December 9, 2019

